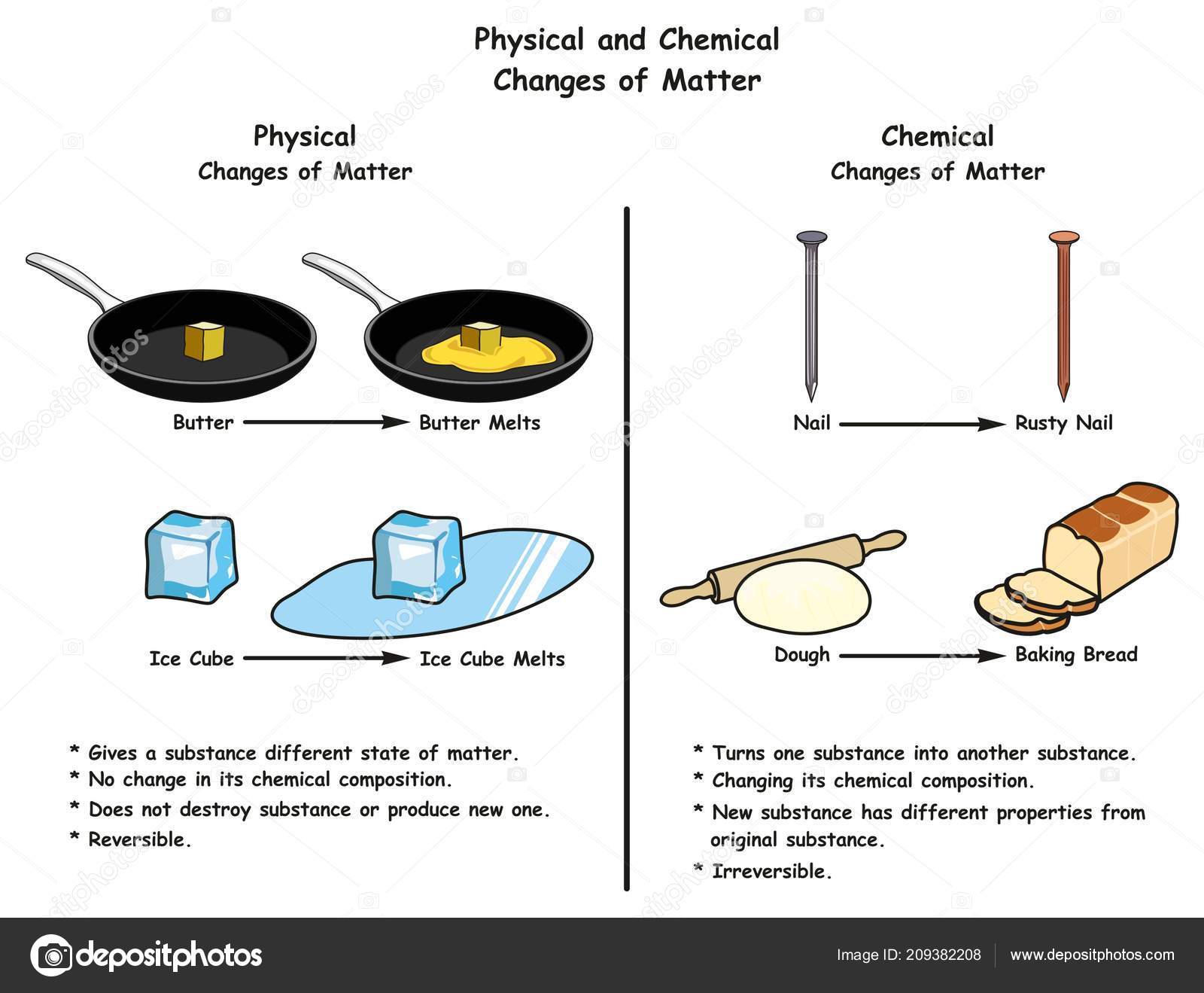
Note: It is important to understand these basic concepts to build up higher level concepts.Ī lot of students get confused here so try to research more about this and not just mugging up definitions.Examples of Physical Changes You See Every DayĪn ice cube melting into water in your drink. Depending upon the nature of the change, these changes can be broadly classified as physical and chemical changes. A process that causes a substance to change into a new substance with a new chemical formula is known as a chemical reaction. The “ingredients” of a reaction are called reactants, and the end results are called products. A process involving the breaking or making of interatomic bonds and the transformation of a substance (or substances) into another. Chemical changes are also known as chemical reactions. Physical changes only change the appearance of a substance, not its chemical composition.Ĭhemical changes cause a substance to change into an entirely substance with a new chemical formula. A few examples of chemical change are digestion of food, burning of coal, rusting, etc Another 30 more examples of chemical and physical change are listed in the table below. As for burning paper, the papers color turns from white to black. The color and smell of a substance change. As in the reaction of water and sodium, it explodes. Some examples of physical change are freezing of water, melting of wax, boiling of water, etc. Production of electrical, sound, and mechanical energy. 
A process that does not cause a substance to become a fundamentally different substance. Physical changes only change the appearance of a substance, not its chemical composition. Original substance cannot be obtained by simple physical method The original form of substance can be regained by simple physical method Very little or no energy in the form of heat, light or sound is usually absorbed or given out in a physical changeĪ chemical change is always accompanied by absorption or evolution of energy No new substance is formed in a physical change.Ī new substance is formed in a physical change. The chemical composition and chemical properties undergo a change 
changed and chemical properties remain unchanged Only physical changes like odour, physical state, density, volume etc.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
